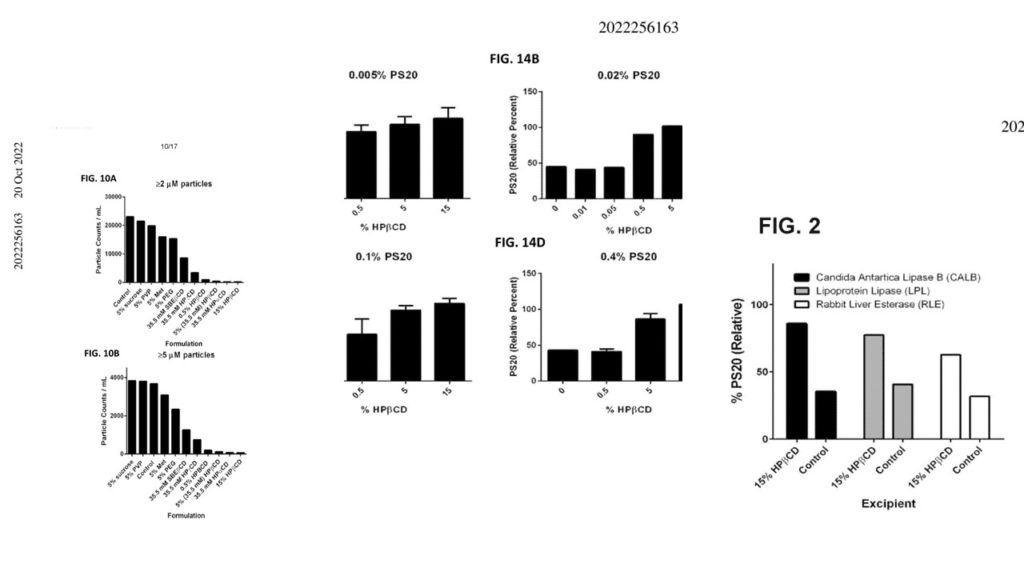
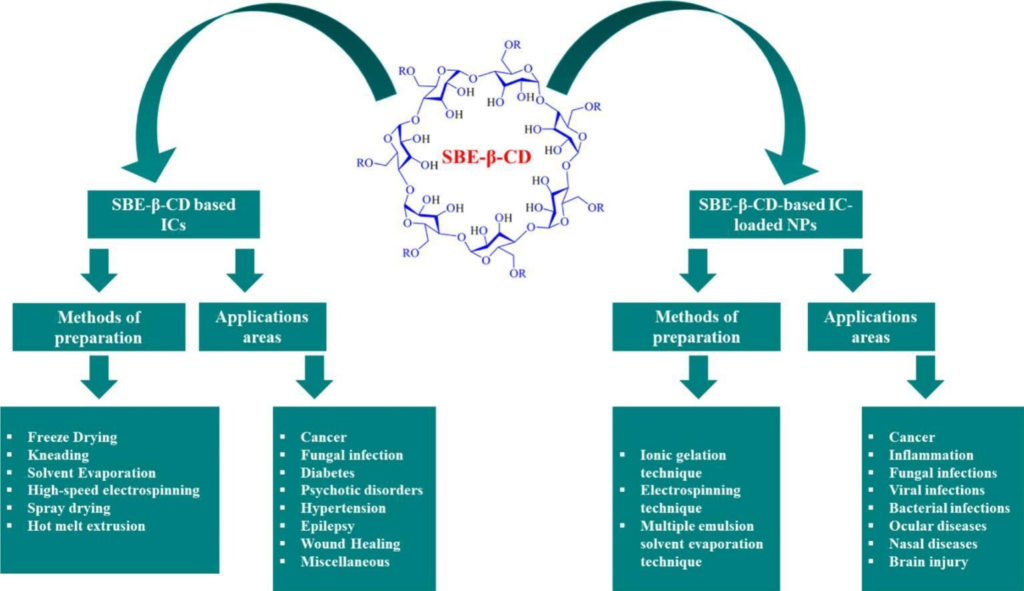
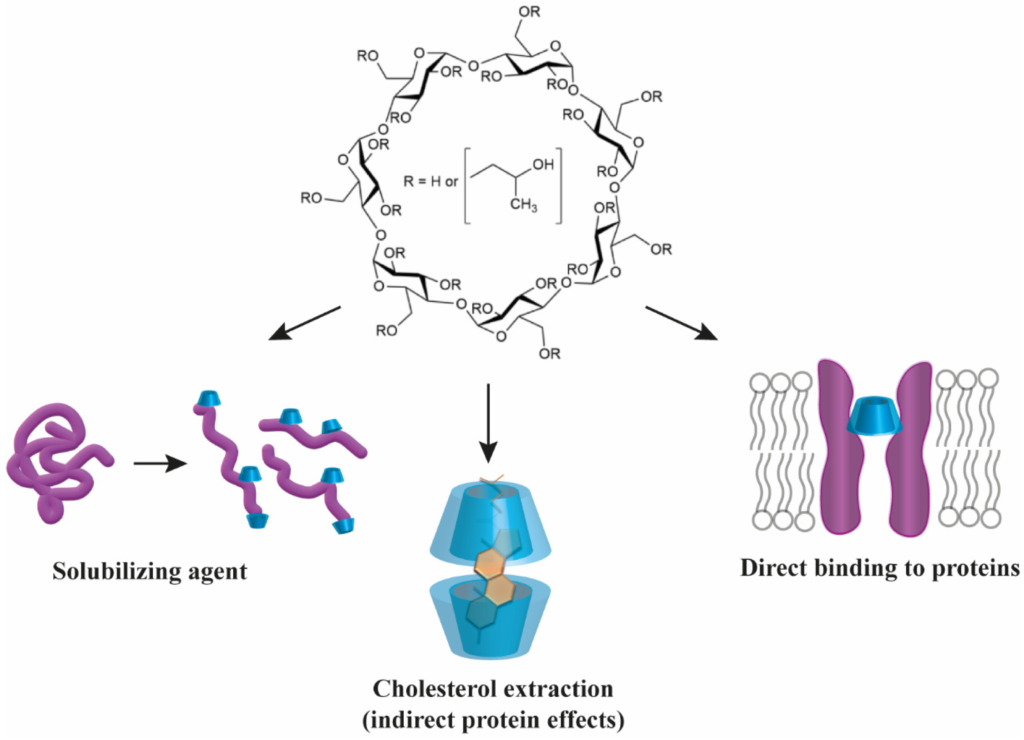
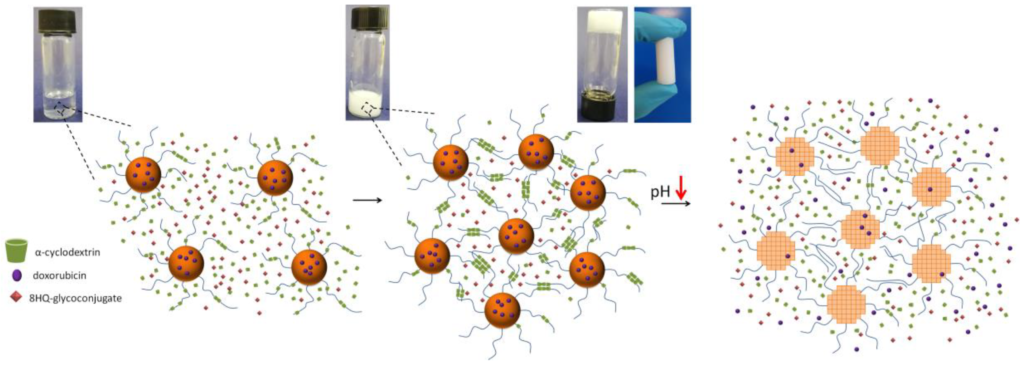
Comparison of the Efficacy and Safety of Adamgammadex with Sugammadex for Reversal of Rocuronium-Induced Neuromuscular Block: Results of a Phase II Clinical Trial
The success of Sugammadex inspired several further cyclodextrin research, including the development of other antidotes and discovering “follow-up” compounds for the same indication. The most advanced candidate in this field is Adamgammadex (developed by Adamerck) with its efficacy recently compared to SGM in a Phase II trial lead by Sichuan University. Although it was used in 2-3 times higher concentrations, the previously reported adverse effects were not observed in this study, including anaphylaxis, haemorrhage, recurarization, abnormal basic vital signs, or lengthened QRS intervals and QT intervals. Adamgammadex was found to be effective for reversing rocuronium-induced neuromuscular block as sugammadex.