Improving the therapeutic window of anticancer agents by β-cyclodextrin encapsulation: Experimental and theoretical insights
Today’s cyclodextrin is about the stabilization effect of CDs on proteins, yet from a different angle.
I shared a lot earlier about the potential of CDs replacing surface active agents, yet how about combining them?
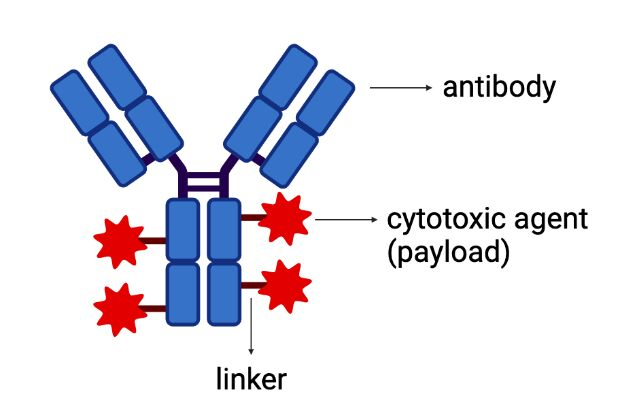
This study from Roquette led by Rajeev Gokhale and Vinod T. investigates the collaborative impact of combining polysorbates and HPβCD as excipients in protein formulations. The introduction of HPβCD in formulations showed it considerably reduced aggregation in two model proteins, bevacizumab and ipilimumab, following exposure to various stress conditions. The diffusion interaction parameter revealed a reduction in protein-protein interactions by HPβCD.
The study suggests that combining these excipients can improve mAb stability in formulations, offering an alternative for the biopharmaceutical industry.