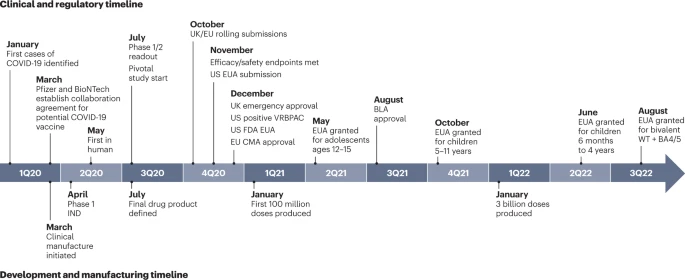
FDA Approves First Respiratory Syncytial Virus (RSV) Vaccine
Today, the U.S. FDA approved Arexvy, the first respiratory syncytial virus (RSV) vaccine approved for use in the United States. Arexvy is approved for the prevention of lower respiratory tract disease caused by RSV in individuals 60 years of age and older. RSV is a highly contagious virus that causes infections of the lungs and breathing passages in individuals of all age groups. RSV circulation is seasonal, typically starting during the fall and peaking in the winter. In older adults, RSV is a common cause of lower respiratory tract disease, which affects the lungs and can cause life-threatening pneumonia and bronchiolitis (swelling of the small airway passages in the lungs). According to the U.S. Centers for Disease Control and Prevention, each year in the U.S., RSV leads to approximately 60,000-120,000 hospitalizations and 6,000-10,000 deaths among adults 65 years of age and older.
GSK’s RSVPreF3 (Arexvy), is a recombinant product that contains a glycoprotein antigen based on the RSV A subgroup and is given with a proprietary adjuvant—the same one in GSK‘s Shingrix shingles (herpes zoster) vaccine—designed to boost the immune response.
See the FDA annucement here